
Blood–brain-barrier organoids for investigating the permeability of CNS therapeutics
Blood–brain-barrier organoids for investigating the permeability of CNS therapeutics
Nature Protocolsvolume 13, pages2827–2843 (2018)
Sonja Bergmann, Sean E. Lawler, Yuan Qu, Colin M. Fadzen, Justin M. Wolfe, Michael S. Regan, Bradley L. Pentelute, Nathalie Y. R. Agar & Choi-Fong Cho
Abstract
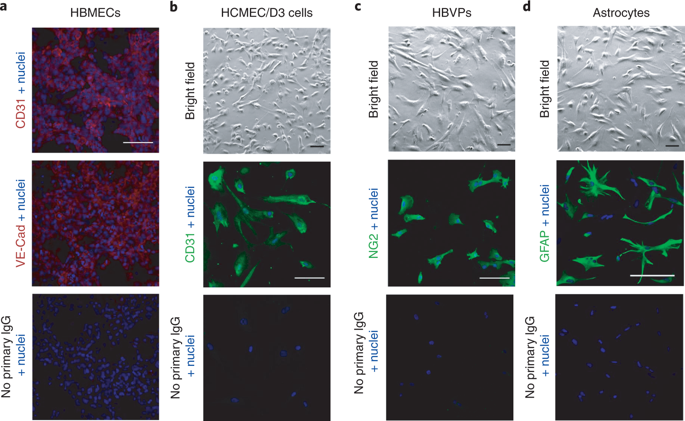
In vitro models of the blood–brain barrier (BBB) are critical tools for the study of BBB transport and the development of drugs that can reach the CNS. Brain endothelial cells grown in culture are often used to model the BBB; however, it is challenging to maintain reproducible BBB properties and function. ‘BBB organoids’ are obtained following coculture of endothelial cells, pericytes and astrocytes under low-adhesion conditions. These organoids reproduce many features of the BBB, including the expression of tight junctions, molecular transporters and drug efflux pumps, and hence can be used to model drug transport across the BBB. This protocol provides a comprehensive description of the techniques required to culture and maintain BBB organoids. We also describe two separate detection approaches that can be used to analyze drug penetration into the organoids: confocal fluorescence microscopy and mass spectrometry imaging. Using our protocol, BBB organoids can be established within 2–3 d. An additional day is required to analyze drug permeability. The BBB organoid platform represents an accurate, versatile and cost-effective in vitro tool. It can easily be scaled to a high-throughput format, offering a tool for BBB modeling that could accelerate therapeutic discovery for the treatment of various neuropathologies.



