
A chemoselective strategy for late-stage functionalization of complex small molecules with polypeptides and proteins
A chemoselective strategy for late-stage functionalization of complex small molecules with polypeptides and proteins
Nature Chemistryvolume 11, pages78–85 (2019)
Daniel T. Cohen, Chi Zhang, Colin M. Fadzen, Alexander J. Mijalis, Liana Hie, Kenneth D. Johnson, Zachary Shriver, Obadiah Plante, Scott J. Miller, Stephen L. Buchwald & Bradley L. Pentelute
Abstract
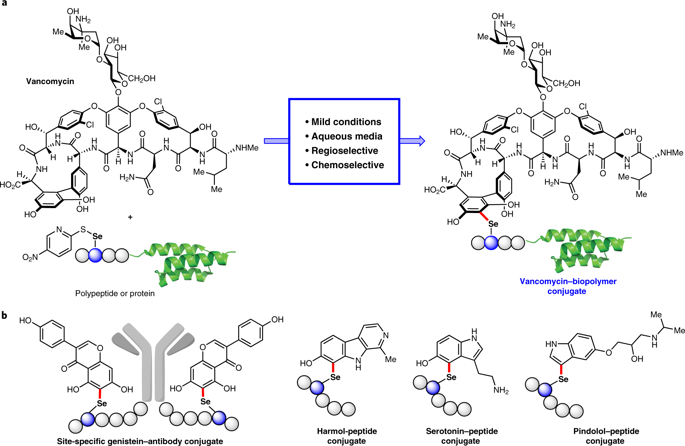
Conjugates between proteins and small molecules enable access to a vast chemical space that is not achievable with either type of molecule alone; however, the paucity of specific reactions capable of functionalizing proteins and natural products presents a formidable challenge for preparing conjugates. Here we report a strategy for conjugating electron-rich (hetero)arenes to polypeptides and proteins. Our bioconjugation technique exploits the electrophilic reactivity of an oxidized selenocysteine residue in polypeptides and proteins, and the electron-rich character of certain small molecules to provide bioconjugates in excellent yields under mild conditions. This conjugation chemistry enabled the synthesis of peptide–vancomycin conjugates without the prefunctionalization of vancomycin. These conjugates have an enhanced in vitro potency for resistant Gram-positive and Gram-negative pathogens. Additionally, we show that a 6 kDa affibody protein and a 150 kDa immunoglobulin-G antibody could be modified without diminishing bioactivity.



