
Salt Effect Accelerates Site-Selective Cysteine Bioconjugation
Salt Effect Accelerates Site-Selective Cysteine Bioconjugation
ACS Cent. Sci., 2016, 2 (9), pp 637–646
DOI: 10.1021/acscentsci.6b00180
Publication Date (Web): August 25, 2016
Peng Dai, Chi Zhang, Matthew Welborn, James J. Shepherd, Tianyu Zhu, Troy Van Voorhis, and Bradley L. Pentelute*
Abstract
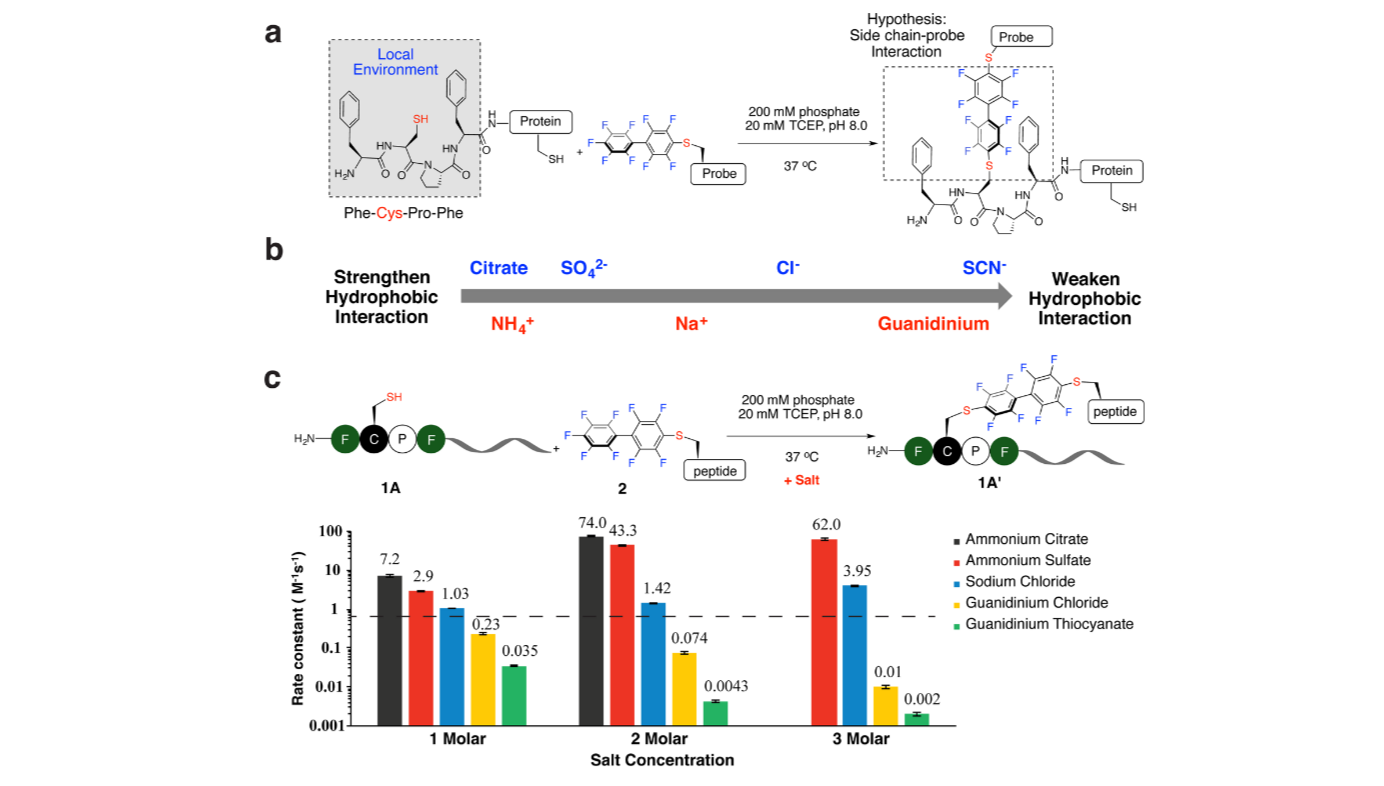
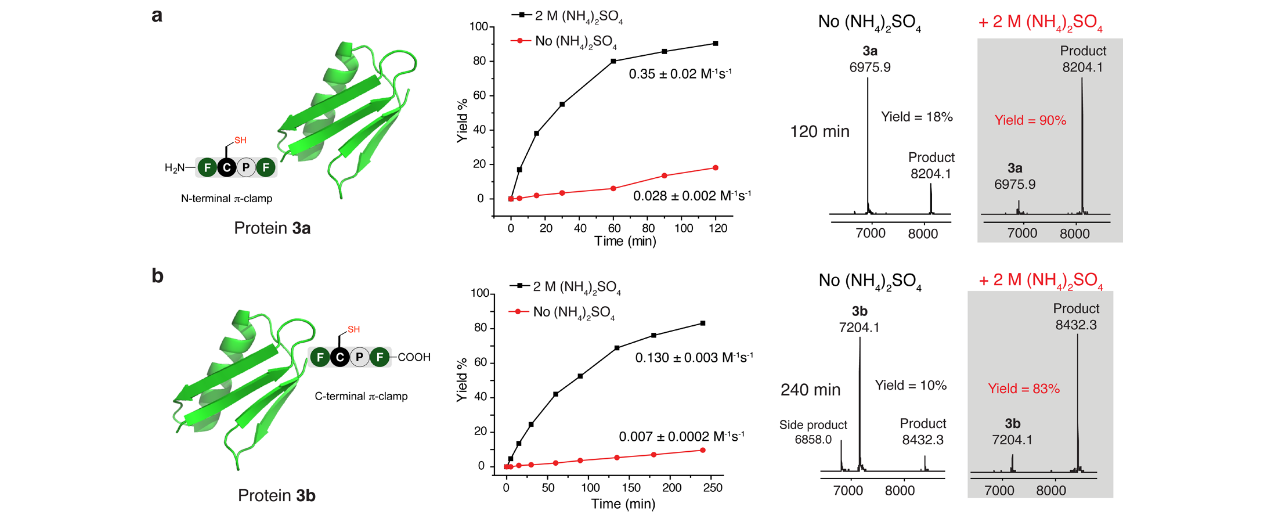
Highly efficient and selective chemical reactions are desired. For small molecule chemistry, the reaction rate can be varied by changing the concentration, temperature, and solvent used. In contrast for large biomolecules, the reaction rate is difficult to modify by adjusting these variables because stringent biocompatible reaction conditions are required. Here we show that adding salts can change the rate constant over 4 orders of magnitude for an arylation bioconjugation reaction between a cysteine residue within a four-residue sequence (π-clamp) and a perfluoroaryl electrophile. Biocompatible ammonium sulfate significantly enhances the reaction rate without influencing the site-specificity of π-clamp mediated arylation, enabling the fast synthesis of two site-specific antibody–drug conjugates that selectively kill HER2-positive breast cancer cells. Computational and structure–reactivity studies indicate that salts may tune the reaction rate through modulating the interactions between the π-clamp hydrophobic side chains and the electrophile. On the basis of this understanding, the salt effect is extended to other bioconjugation chemistry, and a new regioselective alkylation reaction at π-clamp cysteine is developed.



