
Palladium Oxidative Addition Complexes for Peptide and Protein Cross-linking
Palladium Oxidative Addition Complexes for Peptide and Protein Cross-linking
J. Am. Chem. Soc., 2018, 140 (8), pp 3128–3133
DOI: 10.1021/jacs.8b00172
Publication Date (Web): February 6, 2018
Koji Kubota‡, Peng Dai‡ , Bradley L. Pentelute* , and Stephen L. Buchwald*
Abstract
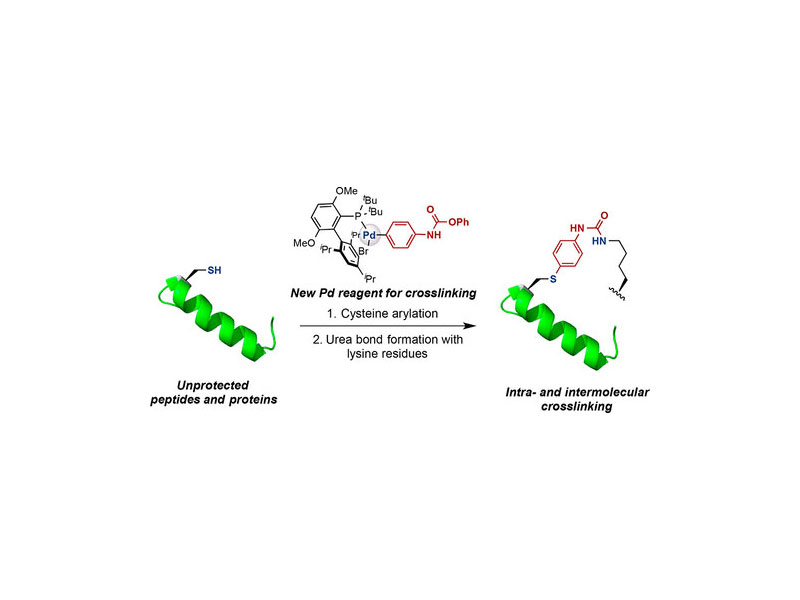
A new method for cysteine–lysine cross-linking in peptides and proteins using palladium oxidative addition complexes is presented. First, a biarylphosphine-supported palladium reagent is used to transfer an aryl group bearing an O-phenyl carbamate substituent to a cysteine residue. Next, this carbamate undergoes chemoselective acyl substitution by a proximal lysine to form a cross-link. The linkage so formed is stable toward acid, base, oxygen, and external thiol nucleophiles. This method was applied to cross-link cysteine with nearby lysines in sortase A*. Furthermore, we used this method for the intermolecular cross-linking between a peptide and a protein based on the p53-MDM2 interaction. These studies demonstrate the potential for palladium-mediated methods to serve as a platform for the development of future cross-linking techniques for peptides and proteins with natural amino acid residues.



