
Cell-Penetrating d-Peptides Retain Antisense Morpholino Oligomer Delivery Activity
Cell-Penetrating d-Peptides Retain Antisense Morpholino Oligomer Delivery Activity
CS Bio Med Chem Au 2022, 2, 2, 150–160, Publication Date:February 16, 2022
Carly K. Schissel, Charlotte E. Farquhar, Annika B. Malmberg, Andrei Loas, and Bradley L. Pentelute
Abstract
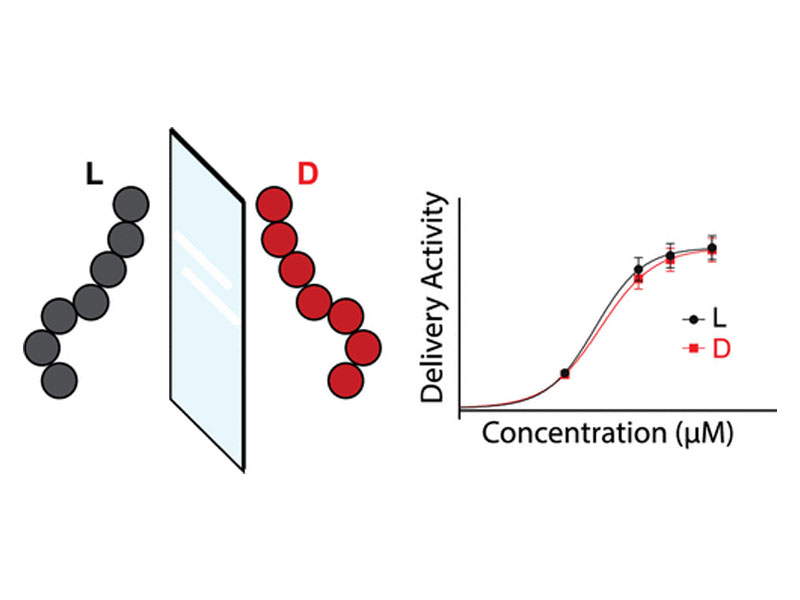
Cell-penetrating peptides (CPPs) can cross the cell membrane to enter the cytosol and deliver otherwise nonpenetrant macromolecules such as proteins and oligonucleotides. For example, recent clinical trials have shown that a CPP attached to phosphorodiamidate morpholino oligomers (PMOs) resulted in higher muscle concentration, increased exon skipping, and dystrophin production relative to another study of the PMO alone in patients of Duchenne muscular dystrophy. Therefore, effective design and the study of CPPs could help enhance therapies for difficult-to-treat diseases. So far, the study of CPPs for PMO delivery has been restricted to predominantly canonical l-peptides. We hypothesized that mirror-image d-peptides could have similar PMO delivery activity as well as enhanced proteolytic stability, facilitating their characterization and quantification from biological milieu. We found that several enantiomeric peptide sequences could deliver a PMO–biotin cargo with similar activities while remaining stable against serum proteolysis. The biotin label allowed for affinity capture of fully intact PMO–peptide conjugates from whole-cell and cytosolic lysates. By profiling a mixture of these constructs in cells, we determined their relative intracellular concentrations. When combined with PMO activity, these concentrations provide a new metric for delivery efficiency, which may be useful for determining which peptide sequence to pursue in further preclinical studies.



